The chemical element berkelium is classed as an actinide metal. It was discovered in 1949 by Stanley Thompson, Albert Ghiorso and Glenn Seaborg.

Data Zone
| Classification: | Berkelium is an actinide metal |
| Color: | silvery-white |
| Atomic weight: | (247), no stable isotopes |
| State: | solid |
| Melting point: | 986 oC, 1259.2 K |
| Boiling point: | 2900 oC, 3173 K |
| Electrons: | 97 |
| Protons: | 97 |
| Neutrons in most abundant isotope: | 150 |
| Electron shells: | 2,8,18,32,27,8,2 |
| Electron configuration: | [Rn] 5f9 7s2 |
| Density @ 20oC: | 14.79 g/cm3 |
Compounds, Radii, Conductivities
| Atomic volume: | 16.7 cm3/mol |
| Structure: | double hexagonal close packed |
| Hardness: | mohs |
| Specific heat capacity | – |
| Heat of fusion | – |
| Heat of atomization | – |
| Heat of vaporization | – |
| 1st ionization energy | 601 kJ mol-1 |
| 2nd ionization energy | – |
| 3rd ionization energy | – |
| Electron affinity | – |
| Minimum oxidation number | 0 |
| Min. common oxidation no. | 0 |
| Maximum oxidation number | 4 |
| Max. common oxidation no. | 3 |
| Electronegativity (Pauling Scale) | 1.3 |
| Polarizability volume | 22.7 Å3 |
| Reaction with air | |
| Reaction with 15 M HNO3 | |
| Reaction with 6 M HCl | |
| Reaction with 6 M NaOH | |
| Oxide(s) | BkO, BkO2, BkO3, Bk2O3 |
| Hydride(s) | BkH2, BkH3 |
| Chloride(s) | BkCl3 |
| Atomic radius | 170 pm |
| Ionic radius (1+ ion) | – |
| Ionic radius (2+ ion) | – |
| Ionic radius (3+ ion) | 96 pm |
| Ionic radius (1- ion) | – |
| Ionic radius (2- ion) | – |
| Ionic radius (3- ion) | – |
| Thermal conductivity | 10 W m-1 K-1 |
| Electrical conductivity | – |
| Freezing/Melting point: | 986 oC, 1259.2 K |
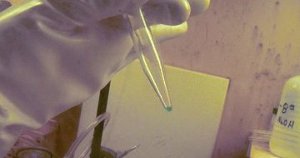
Berkelium-249, contained in the green solution in the tip of the vial, was used in the discovery of element 117, ununseptium. Image Ref.(5)
Discovery of Berkelium
Berkelium was the fifth synthetic transuranium element of the actinide series to be discovered.
It was first produced in 1949 by Stanley Thompson, Albert Ghiorso and Glenn Seaborg in California, USA.
The scientists bombarded 7 milligrams of americium-241 with alpha particles in the 60-inch cyclotron in Berkeley, California. (1), (2)
Each nuclear reaction produced berkelium-243 (half-life 4.5 hours) and two neutrons. (2)
The new element was separated and identified chemically by a combination of precipitation methods and high temperature ion-exchange columns. (2)
Berkelium was isolated in greater quantities for the first time by Burris Cunningham and Stanley Thompson in 1958 at the Materials Testing Reactor in Arco, Idaho by prolonged (five years) neutron irradiation of plutonium-239. Approximately 0.6 micrograms of berkelium and 1.2 micrograms of californium were synthesized. (3)
The first bulk (>1 mg) samples of berkelium metal were prepared in early 1969 by the reduction of BkF3 with lithium metal vapor at about 1300 K. (4)
The element was named after the city of Berkeley, California where it was first produced. The choice of element name was in keeping with some of the other actinides, whose names follow the pattern of the lanthanide elements directly above them in the periodic table.
The lanthanide above berkelium is terbium, named after the place of its discovery, Ytterby, a village in Sweden.


Aerial View of the High Flux Isotope Reactor at Oak Ridge National Laboratory where berkelium has been produced.
Appearance and Characteristics
Harmful effects:
Berkelium is harmful due to its radioactivity.
Characteristics:
Berkelium is a silvery-white highly radioactive metal.
It is chemically reactive, oxidizing slowly in air.
The oxidation may be slow because of the initial formation of a thin protective oxide layer. (4a)
Berkelium metal is dimorphic: double hexagonal close packed at room temperature and face centered cubic at higher temperatures. (4b)
The metal dissolves rapidly in aqueous mineral acids, liberating hydrogen gas and forming Bk (III) in solution. (4c)
Bk (III) ions are green in most mineral acid solutions.
Bk (IV) ions are yellow in HCl solution. (4d)
Uses of Berkelium
Berkelium is of scientific research interest only.
Berkelium-249 is used as a target material for the production of heavier actinides such as lawrencium-260. (4e)
Abundance and Isotopes
Abundance earth’s crust: 0 parts per million by weight, 0 parts per million by moles
Abundance solar system: 0 parts per billion by weight, 0 parts per billion by moles
Cost, pure: $ per g
Cost, bulk: $ per 100g
Source: Berkelium is produced in miniscule amounts in nuclear reactors such as the High Flux Isotope Reactor at Oak Ridge National Laboratory, Tennessee by nuclear bombardment of plutonium, curium or americium with alpha particles.
Isotopes: Berkelium has 12 isotopes whose half-lives are known, with mass numbers 232 to 247. Berkelium has no naturally occurring isotopes. Its longest lived isotopes are 247Bk, with a half-life of 1380 years, 249Bk with a half-life of 320 days and 245Bk with a half-life of 4.94 days.

References
- John Emsley, Nature’s building blocks: an A-Z guide to the elements., Oxford University Press, 2003 p53.
- Glenn T. Seaborg, Modern alchemy: selected papers of Glenn T. Seaborg.,World Scientific, 1994, page 789.
- B. B. Cunningham, Berkelium and Californium., Journal of Chemical Education, Vol 36.1 (1959) p35.
- David E. Hobart and Joseph R. Peterson, The Chemistry of the Actinide and Transactinide Elements., Springer., Vol 3.10, p1457; 4a. p1460; 4b. p1458; 4c. p1461; 4d. p1472; 4e. p1447.
- Photo: ORNL
Cite this Page
For online linking, please copy and paste one of the following:
<a href="https://www.chemicool.com/elements/berkelium.html">Berkelium</a>
or
<a href="https://www.chemicool.com/elements/berkelium.html">Berkelium Element Facts</a>
To cite this page in an academic document, please use the following MLA compliant citation:
"Berkelium." Chemicool Periodic Table. Chemicool.com. 15 Oct. 2012. Web. <https://www.chemicool.com/elements/berkelium.html>.
This was very helpful, thank you.
The only thing I would ask is an example of how you would like to be cited in APA format.
Hi Trinity, I think it would be (but no guarantees!):
Chemicool. (2012). Berkelium element facts. Retrieved Oct. 15, 2012, from http://www.chemicool.com/elements/berkelium.html