Anisotropy is the property of substances to exhibit variations in physical properties along different molecular axes. It is seen in crystals, liquid crystals and, less commonly, in liquids.
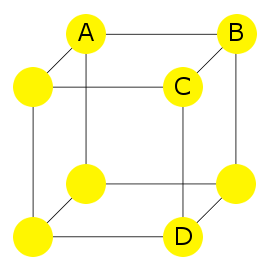
For example, consider the primitive cubic crystal lattice structure shown here. In this instance, all of the atoms are of the same element.
To recognize this structure's anisotropy, consider the distances A-B, A-C and A-D; they are all different.
If the A-B distance is 1 unit, A-C is √2 units, and A-D is √3 units.
Viewing the structure along an axis following the direction A-B looks different from along an axis following the directions A-C or A-D. This leads to different physical and mechanical properties in a single crystal along the different axes: examples are different electrical and thermal conductivity and light polarization.
In amorphous materials, such as glass, no long-range order exists; properties are identical in all directions; these are isotropic materials.
