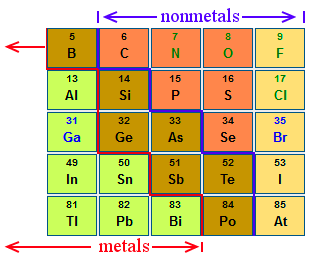
A metalloid is a chemical element that exhibits some properties of metals and some of nonmetals. In the periodic table metalloids form a jagged zone dividing elements that have clear metallic properties from elements that have clear nonmetallic properties.
Definition for metalloids: elements with properties intermediate between metals and nonmetals.
Boron, silicon, germanium, arsenic, antimony, tellurium, and polonium are metalloids. In some cases, authors may also class selenium, astatine, aluminum, and carbon as metalloids, but this is less common.
The reason for differences in classification is that no clear, unambiguous definition exists for metalloids. In the periodic table, the farther you look to the left or right of the metalloid dividing zone, the clearer the metallic or nonmetallic properties become. The closer you get to the zone, the more blurred the properties become.
Metalloids tend to be semiconductors; silicon is the best known example of a semiconductor. Most microchips and microprocessors are made with silicon.
