Interaction of a substance with water is a very general definition of hydration.

More specifically, there are several different situations:
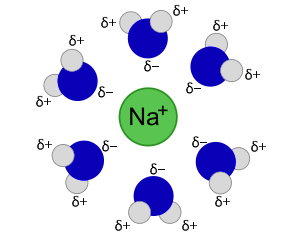
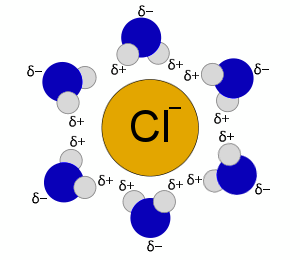
When a salt, such as sodium chloride, dissolves in water, the sodium and chloride ions undergo hydration.
Each individual ion is surrounded by water molecules.
The water molecules are polar, with a slight excess of negative charge on oxygen and a slight excess of positive charge on the hydrogens.
The water's oxygens are oriented towards the sodium through electrostatic attraction, allowing the sodium to dissolve, as shown in the image. Such an arrangement is known as a hydration shell or hydration sphere.
Similarly, a chloride ion would be hydrated with water's slightly positive hydrogen's oriented towards the chloride.
Hydration is an example of solvation.
A hydration reaction is a chemical reaction. Water is one of the reactants that is transformed into products.
One of the commonest hydration reactions is the hydration of alkenes to produce alcohols.
For example, industrial ethanol is manufactured by the hydration of ethene.
It is common for salt crystals to contain water in a definite ratio to the salt. Some salts are hygroscopic, attracting moisture from the atmosphere to their crystals.
For example, dried calcium chloride, CaCl2, left in normal conditions will attract water to form CaCl2(H2O)6. The water molecules form a hydration shell around the calcium ion.