Extractions are a way to separate a desired substance when it is mixed with others. The mixture is brought into contact with a solvent in which the substance of interest is soluble, but the other substances present are insoluble.
Extractions use two immiscible phases (these are phases that do not mix, like oil and water) to separate the substance from one phase into the other.
Easy Example of Extraction
Tea-making is a very basic non-laboratory extraction. You boil tea leaves in water to extract the tannins, theobromine, polyphenols, and caffeine out of the solid tea leaves and into the liquid water. You are then free to drink and enjoy the water and the extracted substances it contains.
Laboratory Extractions
Typical lab extractions are to transport organic compounds out of an aqueous phase and into an organic phase. These are often carried out using a separating funnel, as illustrated below.
The distribution of a solute between two phases is an equilibrium described by partition theory.
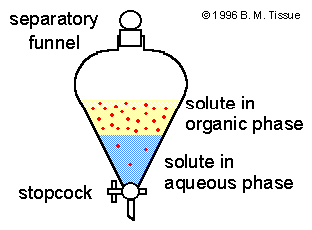
Illustration of an extraction in a separatory funnel
Elemental analysis generally requires fairly simple (not necessarily easy) sample preparation.
Solids are usually dissolved or digested in caustic solution and liquids are sometimes extracted to separate the analyte from interferences.
Organic analysis is often much more complicated. Real-world samples can be very complicated matrices that require careful extraction procedures to obtain the analyte(s) in a form that can be analyzed.

